Ionization Mechanisms in UV-MALDI
Previous: Primary Ionization Table of Contents Next: Rate Equation Model
3b. Secondary Ionization
Table 2, Proton Affinities
Table 3, Cation Affinities
A Remark on IR vs UV MALDI
The Second Ionization Step: Making the analyte ions we observe
The density changes slowly enough in the plume that many collisions
occur. As a result the primary ions can react with the neutral
molecules to give new ions, which are finally detected. This is the second step of the CPCD UV MALDI ionization model, the secondary ionization period. It is much longer than primary ionization. Note that secondary processes start as soon as primary ions exist, so the two steps overlap in time.
Analyte ions are derived from primary matrix ions by one or more of 3 reactions (m=matrix, A=analyte):
The many plume collisions allow approach to local thermal equilibrium. A consequence is that the ions left at the end of the plume expansion are, generally speaking, the thermodynamically most favorable ones. The basic knowledge needed to qualitatively predict and interpret MALDI spectra is therefore the thermodynamics of matrix-analyte and analyte-analyte ion-molecule reactions.
Thermodynamics takes us a long way, but it isnt quite that simple in MALDI. True thermodynamic equilibrium would mean recombination of all ions, they are far too energetic to exist in significant quantities in the plume. So for a quantitative picture we do still need to know something about the kinetics of the main plume reactions. See the positive/negative discussion for an example of kinetic limitations in MALDI.
But what are the reactants exactly? The plume contains clusters, droplets and chunks, but because the plume expands by several orders of magnitude, the last collisions experienced are essentially always bimolecular. Because matrix is present in large excess, it will be the last collision partner for nearly all analyte molecules. This is very helpful, since it means the thermodynamics and kinetics of simple bimolecular reactions are the most important, not what happens in a complicated cascade involving aggregates of many sizes and compositions.
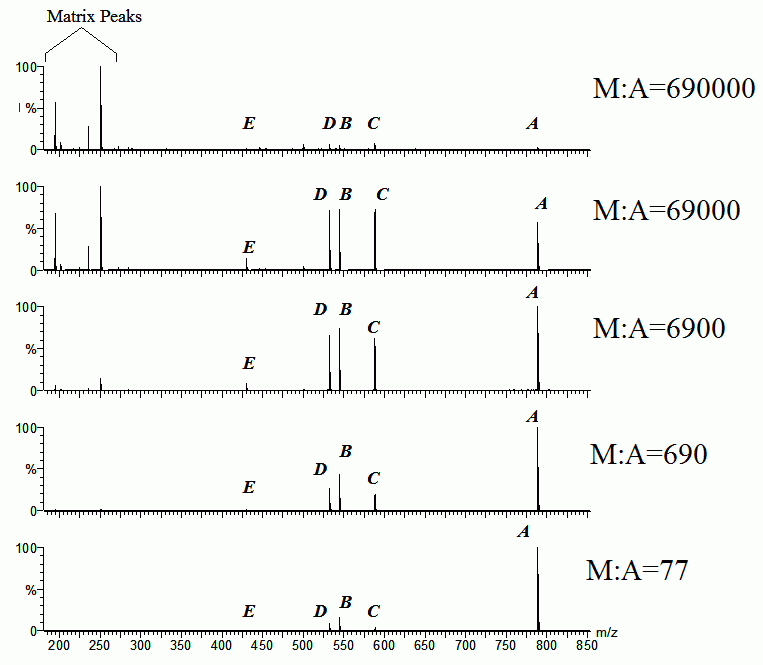
Evidence that matrix and analyte do undergo such reactions is abundant. Very large changes in relative signal strength are observed depending on simple experimental variables like matrix/analyte mole ratio, or laser intensity. An example is shown in Fig. 7.

Figure 7. Example of matrix and analyte suppression effects as a function of matrix to analyte mole ratio. Five analytes (A-E) were present in equimolar amounts, the matrix was DCTB. The indicated mole ratios are for matrix to the sum of all analytes. In the most dilute samples matrix ions are abundant, and the analytes all appear with reasonably consistent relative intensities. As more analyte is added, the matrix signals first disappear, then the other analytes are significantly suppressed by A. See Hoteling, Knochenmuss, et al, Eur. J. Mass Spectrom., vol. 12, p. 345 (2006).
The analytes in Fig. 7 all appear as radical cations, and so are formed by reaction 2 above. Nevertheless, they also suppress protonated and cationed matrix signals. This shows that the various ion types interconvert in the plume. Charge flows to the thermodynamically most favorable species, in this case analyte A. It was shown that this analyte has the lowest ionization potential, it is the least able to retain an electron in an ion-molecule collision.
Suppression of one analyte by another is common in MALDI. If both are radicals or protonated this is easy to understand, but even analytes which preferentially ionize in different ways can suppress each other. An example is shown in Fig. 8, where a protonating species suppresses on that preferentially forms potassium adducts.
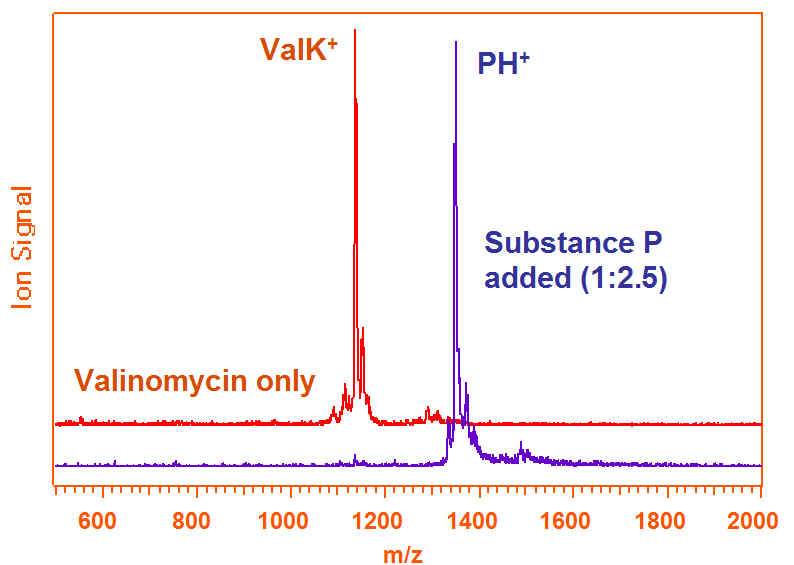
Figure 8. Example of dissimilar analyte ion suppression effects. See Knochenmuss, et al, J. Mass Spectrom., vol. 35, p. 1237 (2000).
Considerable information has become available on the thermodynamics of MALDI secondary ionization reactions. For many biomolecules (e.g. proteins and peptides), protonation reactions are dominant. Peptides with strongly basic residues are well known to be much more easily detected that those without. From the two-step model of secondary reactions this is readily understood. Common matrices have lower proton affinities or gas-phase basicities than do the basic amino acids. Reaction 1 can be exothermic by up 100 kJ/mol or more, a strong driving force.
| Matrix | Proton affinity, kJ/mol |
| 4HCCA | 841, 933±9, 841.5 |
| 2,5-DHB | 855±8, 853.5±16.7, 854±14, 850.4, 855.8 |
| Ferulic acid | 879 |
| Sinapinic acid | 887, 894.5±13.5, 875.9 |
| 3-HPA | 896, 898.5 |
| HABA | 943, 950 |
| Dithranol | 874.5±8.4, 885.53 |
| THAP | 882, 893 |
| Analyte | |
| Gly | 885±13, 859.9, 863 |
| His | 955±9, 969.2, 937 |
| Arg | >1016 |
| Gramicidin S | >1018.0 |
| Bradykinin | >1025.1 |
| Leucine-enkephalin | 967.8±2.1 |
| Cytochrome C | (5H+ ion) 735, (4H+) 722, (3H+) 673 |
For analysis of substances with low proton affinity, such as many synthetic polymers, cationization by metal ions (reaction 3) is often the most favorable ionization mechanism. Because the ions are larger than protons, the affinities are lower, as shown in the Table 3. The reaction exothermicities are also correspondingly lower, perhaps 25 kJ/mol. This means that protonation is a more favorable ionization route than cationization for all analytes with a basic site. It is consistent with the general experience that cationized satellite peaks of protonated analytes are less intense. This is, of course, also modulated by the amount of cation (salt) available.
| Matrix | K+ affinity, kJ/mol | Na+ affinity, kJ/mol | H+ affinity, kJ/mol |
| Sinapinic acid | 104 | 159 | 860 |
| DHB | 99 | 158 | 860 |
| THAP | 97 | 154 | 878 |
| Dithranol | 94 | 150 | 874 |
| Analytes | |||
| dipeptides | >160 | ||
| nucleobases | 165-190 | ||
| carbohydrates | >160 |
Secondary reactions and IR MALDI
As a small aside, the secondary reactions of two step model explain an observation that was initially somewhat puzzling: the similarity of the spectrum of a sample ablated with either IR or UV. There are some differences (e.g. higher charger states are more common in IR), and IR ablates much more sample per observed ion, but the similarities are striking.
The primary processes in IR MALDI are certainly not the same as those explained in the last section for UV. But as this section has discussed, and as will be made quantitative in the next one, secondary reactions go a long way toward the most favorable ions. So even if the primary ions of IR MALDI are completely different than those of UV MALDI, the secondary reactions will lead to the same products. In IR MALDI the secondary reactions are probably even more extensive, because so much material is ablated, and the ion density is lower. In other words, because of secondary reactions IR and UV MALDI must give similar results.
Previous: Primary Ionization Next: Rate Equation Model
MALDI Ionization Tutorial © Copyright 2007-2016 Richard Knochenmuss